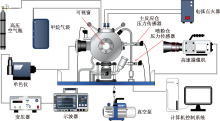
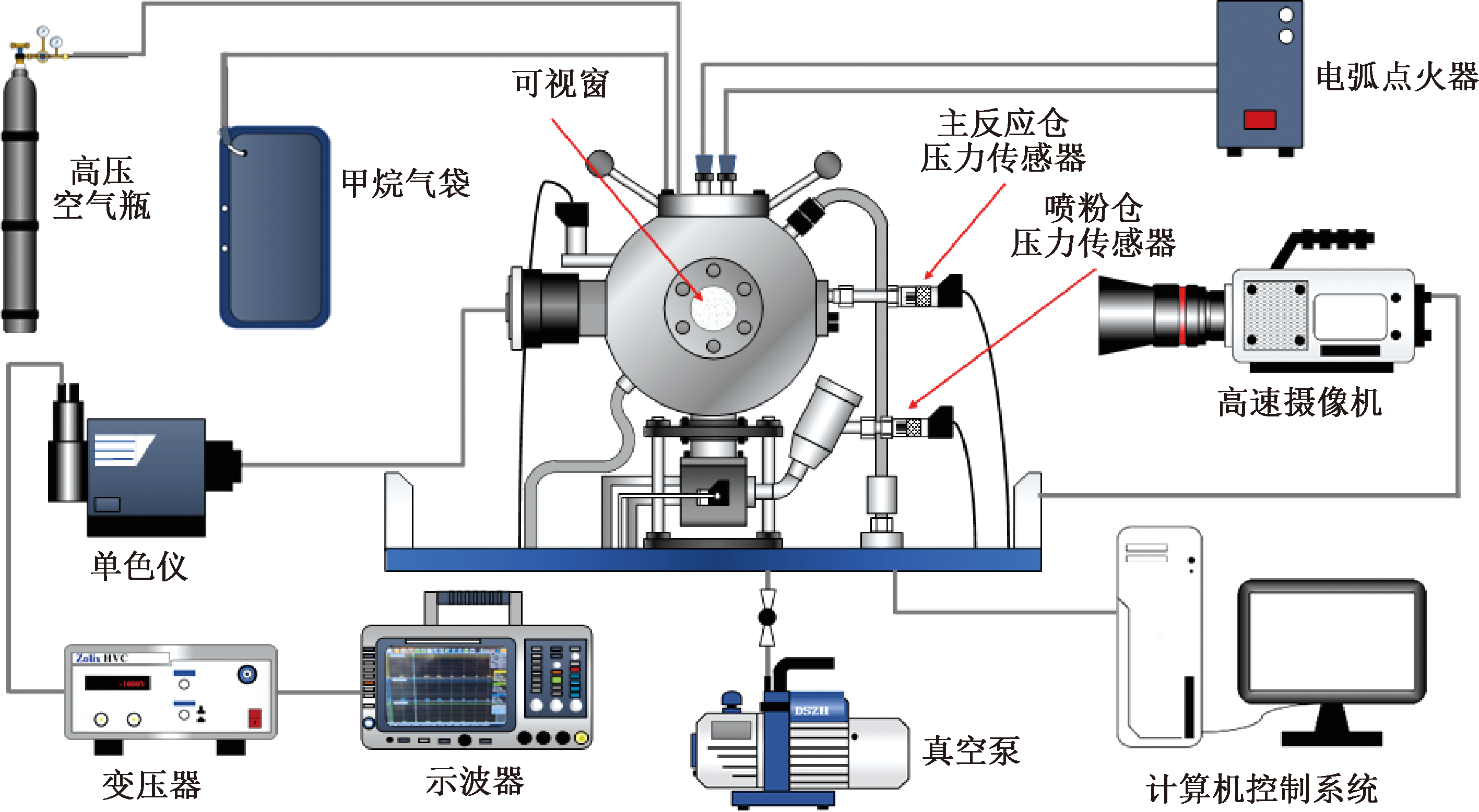
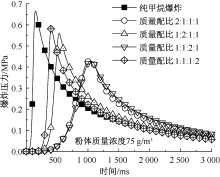
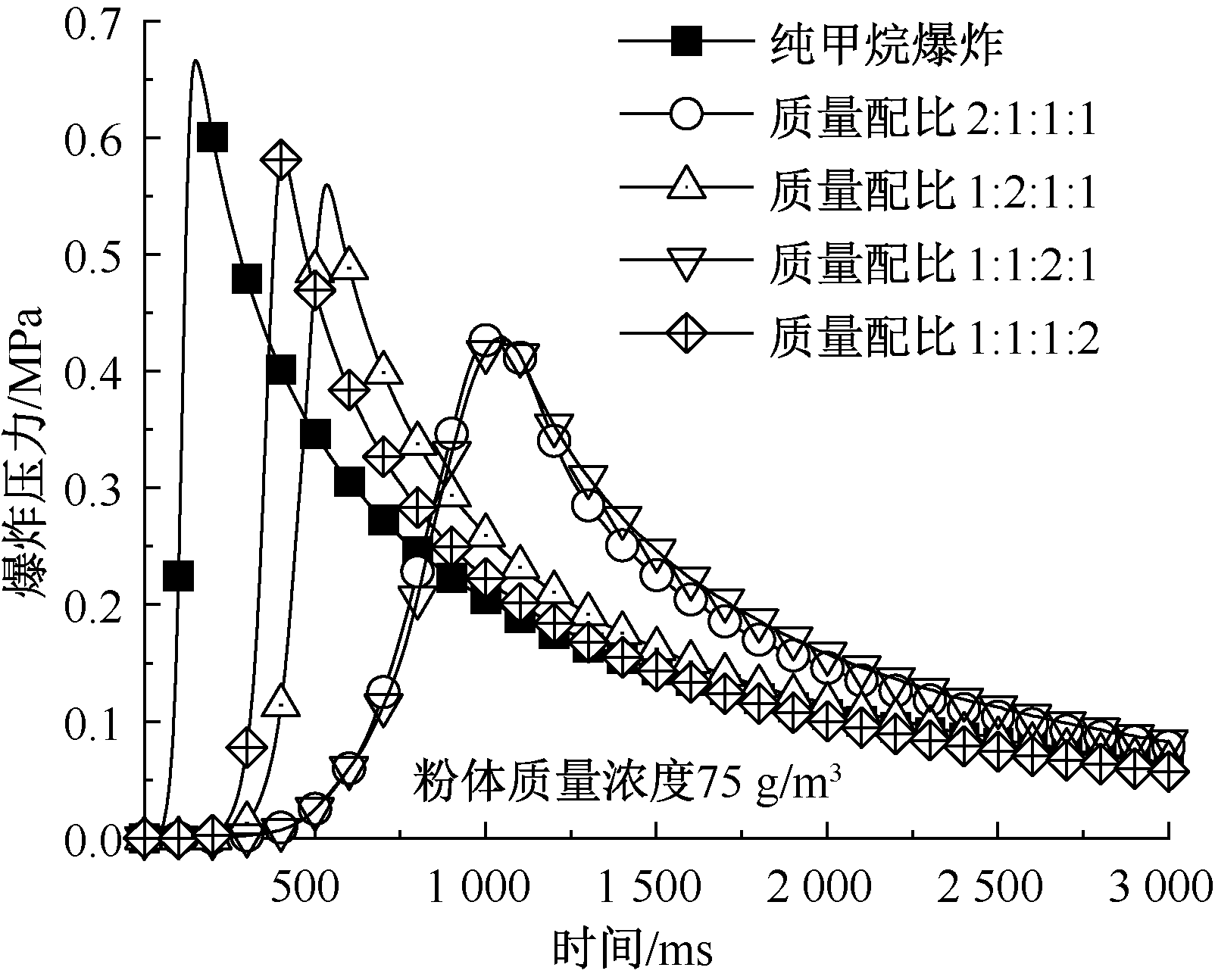
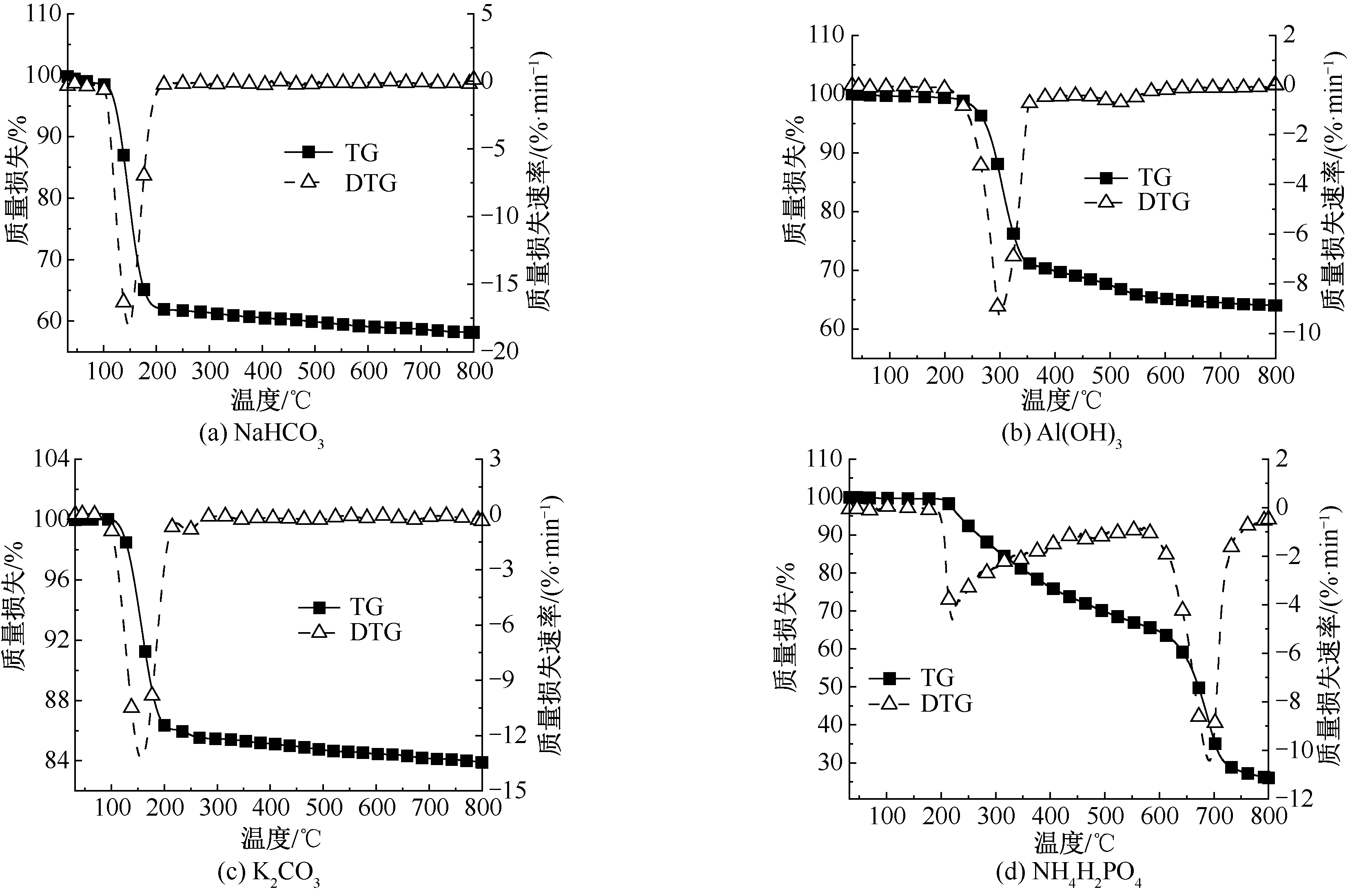
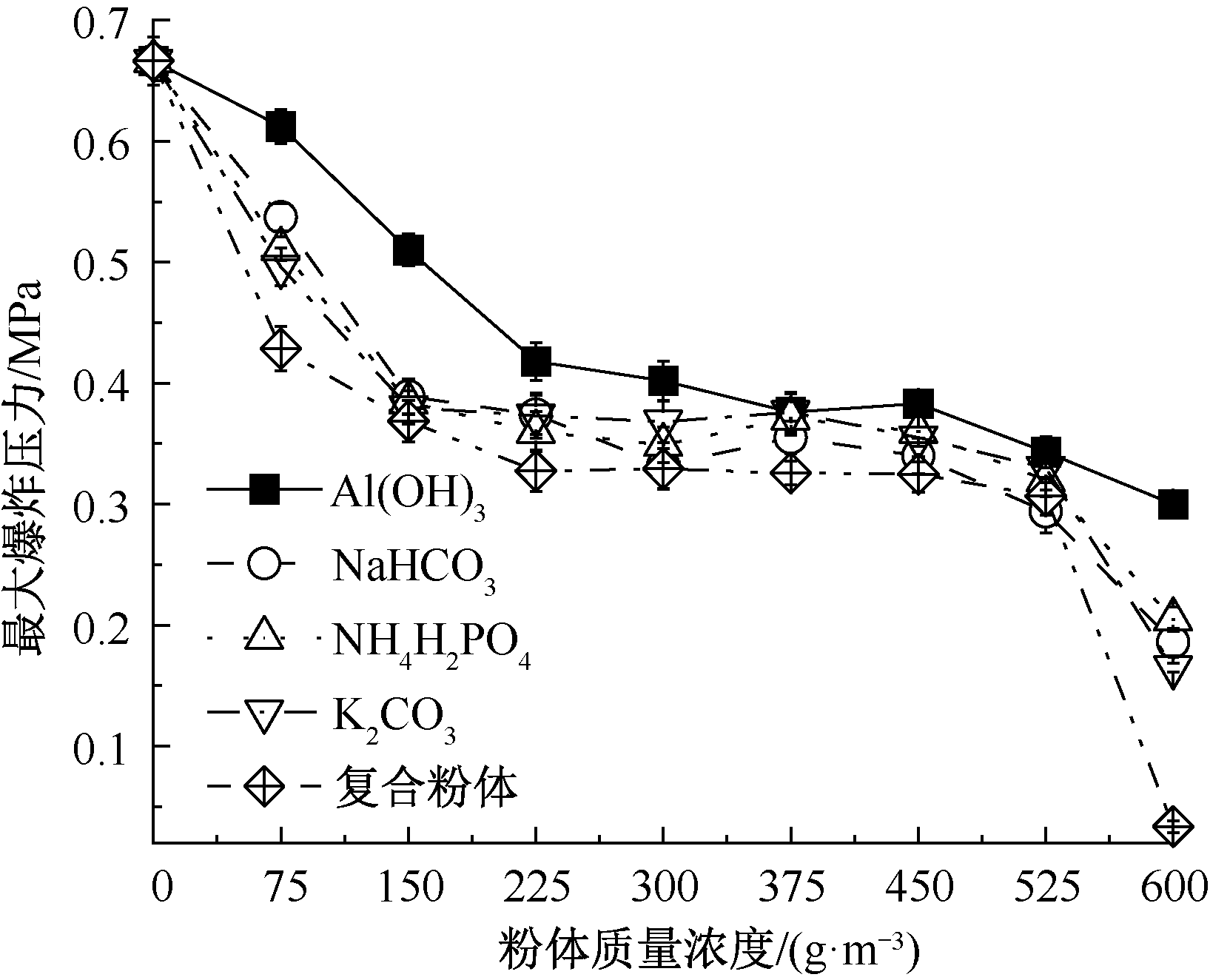
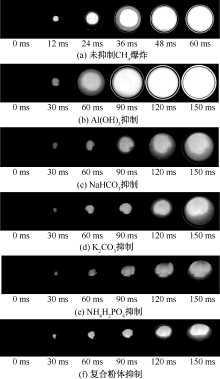
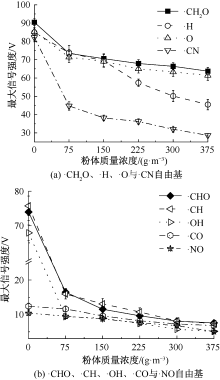
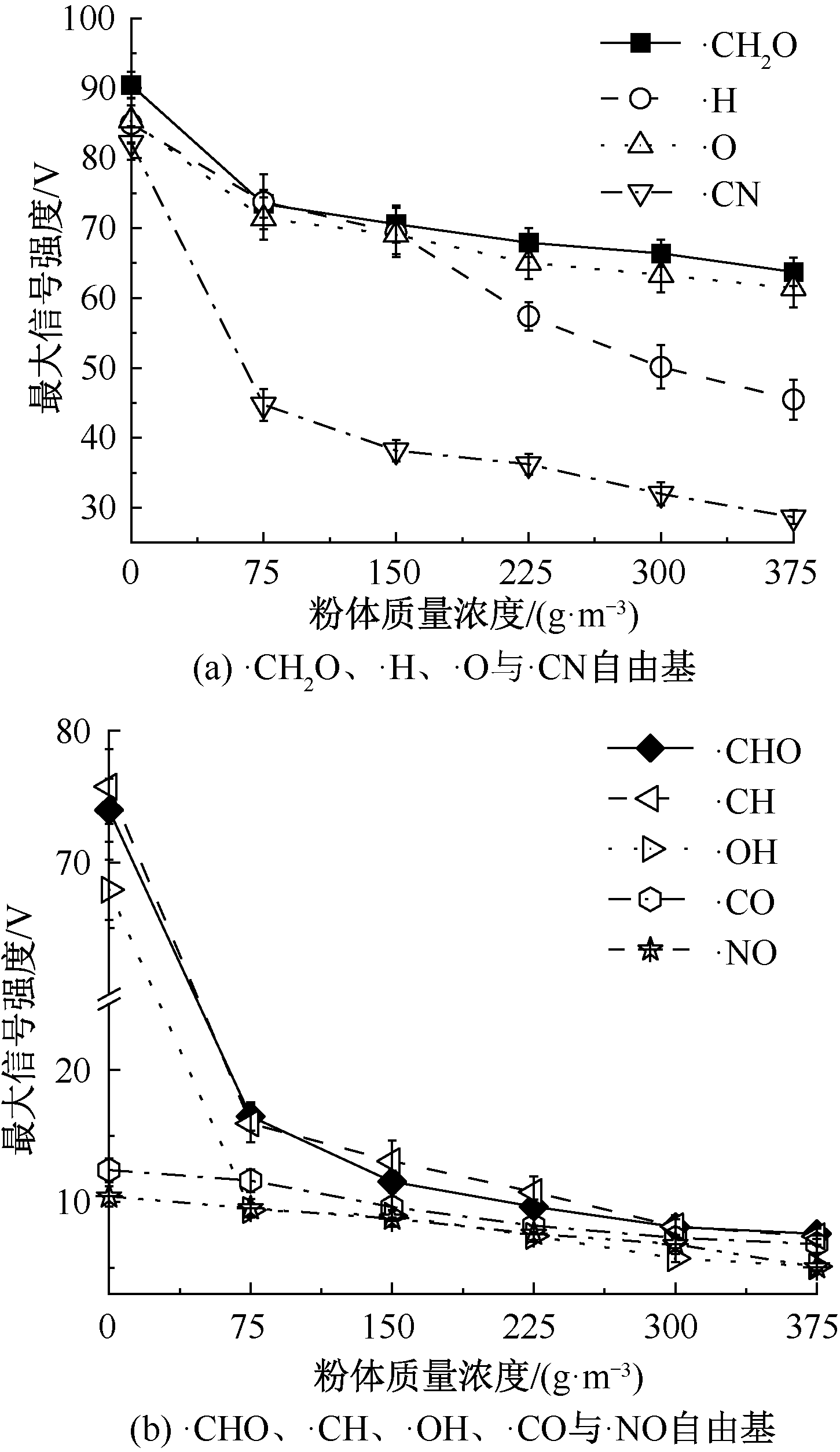
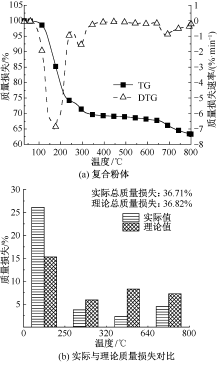
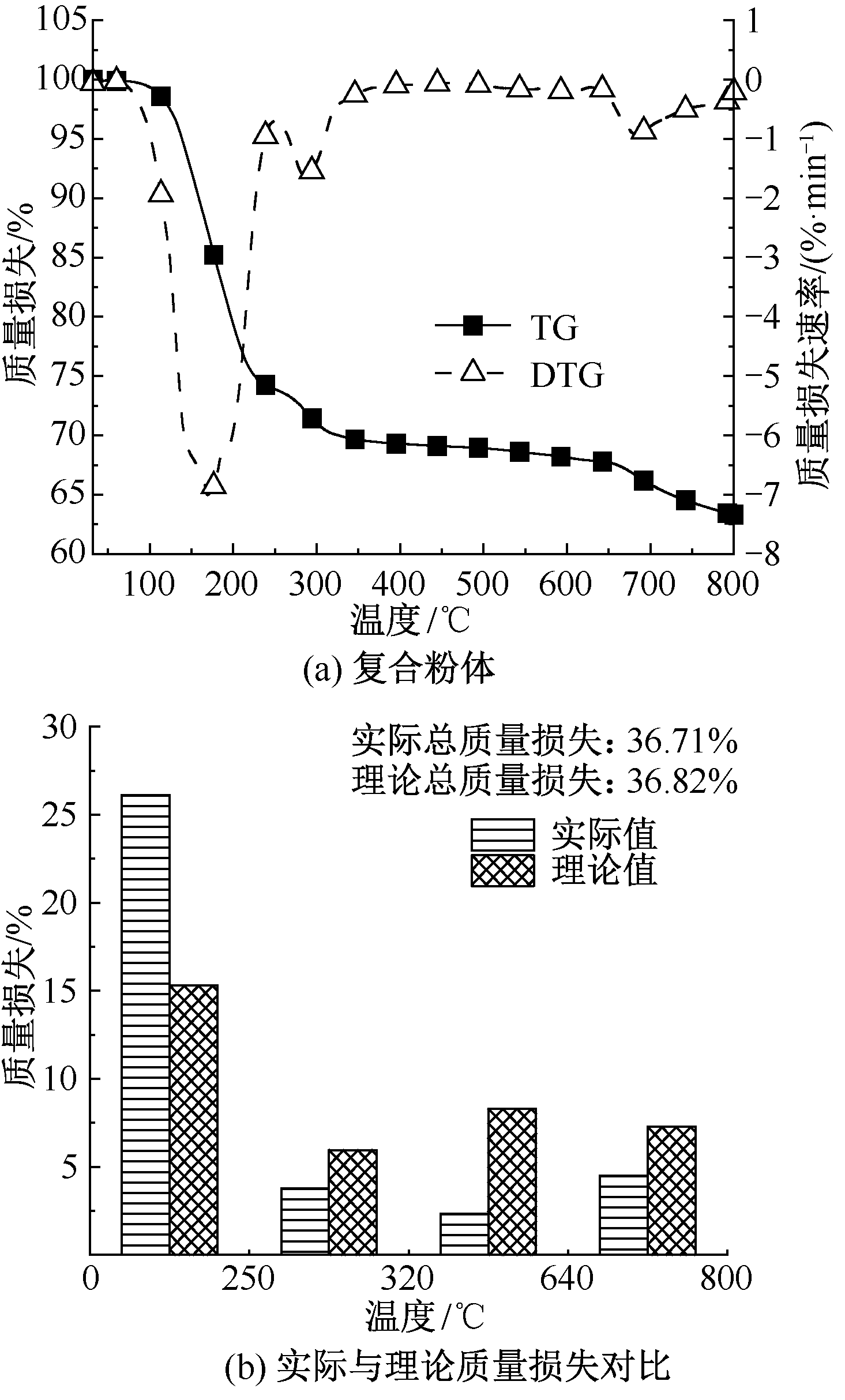
| [1] |
YU Minggao, LI Shanshan, LI Haitao, et al. Numerical evaluation of the influence of initial pressure/temperature on the explosion properties and soot formation of methane/coal volatiles mixtures[J]. Fuel, 2023, 331(1): DOI: 10.1016/j.fuel.2022.125698.
|
| [2] |
张莉聪, 李斯曼, 周振兴. 煤矿瓦斯多相协同抑爆的研究进展与展望[J]. 中国安全科学学报, 2023, 33(增1): 97-104.
|
|
ZHANG Licong, LI Siman, ZHOU Zhenxing. Research progress and prospect of multi-phase synergistic explosion suppression of coal mine gas[J]. China Safety Science Journal, 2023, 33(S1): 97-104.
doi: 10.16265/j.cnki.issn1003-3033.2023.S1.0016
|
| [3] |
李敏, 王丹, 贺珊, 等. 煤矿瓦斯爆炸风险评估研究综述及进展[J]. 中国安全科学学报, 2025, 35(2): 127-136.
doi: 10.16265/j.cnki.issn1003-3033.2025.02.0963
|
|
LI Min, WANG Dan, HE Shan, et al. Research review and progress of coal mine gas explosion risk assessment[J]. China Safety Science Journal, 2025, 35(2): 127-136.
doi: 10.16265/j.cnki.issn1003-3033.2025.02.0963
|
| [4] |
LIANG He, YAN Xingqing, SHI Enhua, et al. Flame evolution and pressure dynamics of premixed stoichiometric ammonia/hydrogen/air in a closed duct[J]. Fuel, 2024, 363(1): DOI: 10.1016/j.fuel.2024.130983.
|
| [5] |
王晓玲, 刘震起. 甲烷-空气预混区外含钾细水雾抑爆特性研究[J]. 中国安全科学学报, 2024, 34(1): 150-157.
doi: 10.16265/j.cnki.issn1003-3033.2024.01.0695
|
|
WANG Xiaoling, LIU Zhenqi. Study on explosion suppression characteristic of water mist containing potassium compounds outside methane-air premixed area[J]. China Safety Science Journal, 2024, 34(1): 150-157.
doi: 10.16265/j.cnki.issn1003-3033.2024.01.0695
|
| [6] |
王燕, 李忠, 张一民, 等. 不同磷酸氢盐作用下的乙烯抑爆特性及机制[J]. 中国安全科学学报, 2022, 32(5): 48-54.
doi: 10.16265/j.cnki.issn1003-3033.2022.05.1564
|
|
WANG Yan, LI Zhong, ZHANG Yimin, et al. Suppression characteristics and mechanism of different hydrogen phosphates on ethylene explosion[J]. China Safety Science Journal, 2022, 32(5): 48-54.
doi: 10.16265/j.cnki.issn1003-3033.2022.05.1564
|
| [7] |
DAI Huaming, LIANG Guangqian, YIN Hepeng, et al. Experimental investigation on the inhibition of coal dust explosion by the composite inhibitor of carbamide and zeolite[J]. Fuel, 2022, 308(2): DOI: 10.1016/j.fuel.2021.121981.
|
| [8] |
ZHU Yujian, MENG Xxiangbao, YU Xiaozhen, et al. Study on inhibition of titanium powder explosion by melamine polyphosphate/iron modified carbon nanotube composite powder[J]. Advanced Powder Technology, 2025, 36(4): DOI: 10.1016/j.apt.2025.104833.
|
| [9] |
张延松, 徐畅, 秦江, 等. 纳米复合抑爆粉体抑制甲烷煤尘混合爆炸动力学机理研究[J]. 安全与环境学报, 2024, 24(11): 4296-4305.
|
|
ZHANG Yansong, XU Chang, QIN Jiang, et al. Study on the dynamic mechanism of nanocomposite explosion suppression powder in mitigating the combined explosion of methane and coal dust[J]. Journal of Safety and Environment, 2024, 24(11): 4296-4305.
|
| [10] |
ZHANG Shuqi, ZHAO Jiangping, YANG Yong, et al. Suppression effect and mechanism of melamine polyphosphate /zinc borate composite suppressant on aluminum dust explosion[J]. Powder Technology, 2025, 456(4): DOI: 10.1016/j.powtec.2025.120777.
|
| [11] |
PAN Rongkun, WANG Chensheng, ZHANG Qingsheng, et al. Experiment and mechanism of explosion suppression thermal properties of multicomponent powder[J]. Thermochimica Acta, 2025, 746(4): DOI: 10.1016/j.tca.2025.179953.
|
| [12] |
LI Manhou, JI Shijie, LI Quan, et al. Study on suppression of ABC powder and potassium bicarbonate-zeolite composite powder on explosion of hydrogen enriched compressed natural gas[J]. International Journal of Hydrogen Energy, 2024, 81(9): DOI: 10.1016/j.ijhydene.2024.07.308.
|
| [13] |
吉冬雨, 杨克, 纪虹, 等. 多巴胺包覆珍珠岩对抑制甲烷爆炸的试验研究[J]. 消防科学与技术, 2024, 43(11): 1505-1511.
|
|
JI Dongyu, YANG Ke, JI Hong, et al. Experimental study of dopamine-coated perlite on inhibition of methane explosion[J]. Fire Science and Technology, 2024, 43(11): 1505-1511.
|
| [14] |
王涛, 孟帆, 弋伟斋, 等. 碳酸钾改性干水-六氟丙烷抑制甲烷爆炸特性[J]. 高压物理学报, 2025, 39(4): 79-90.
|
|
WANG Tao, MENG Fan, YI Weizhai, et al. Coupling inhibition effects of dry water modified by potassium carbonate and hexafluoropropane on methane explosion[J]. Chinese Journal of High Pressure Physics, 2025, 39(4): 79-90.
|
| [15] |
刘丽, 孟祥豹, 陈记合, 等. 含磷回收物对甲烷/煤尘复合爆炸火焰传播的抑制效果及机理研究[J]. 煤矿安全, 2025, 56(3): 75-83.
|
|
LIU Li, MENG Xiangbao, CHEN Jihe, et al. Study on suppression effect and mechanisms of phosphorus-containing recycling substance on flame propagation of methane/coal dust composite explosion[J]. Safety in Coal Mines, 2025, 56(3): 75-83.
|
| [16] |
李孝斌, 张瑞杰, 孙婧雯, 等. 甲烷爆炸中尿素粉体浓度对典型自由基影响研究[J]. 安全与环境学报, 2023, 23(4): 1093-1100.
|
|
LI Xiaobin, ZHANG Ruijie, SUN Jingwen, et al. Experiment of typical free radical change during urea inhibition of methane explosion[J]. Journal of Safety and Environment, 2023, 23(4): 1 093-1 100.
|
| [17] |
张莉聪, 李斯曼. 基于基元反应的气液两相协同抑爆阻燃效果分析[J]. 中国安全科学学报, 2024, 34(3): 101-108.
doi: 10.16265/j.cnki.issn1003-3033.2024.03.1169
|
|
ZHANG Licong, LI Siman. Analysis of gas-liquid two-phase coordinated explosion and flame retardant effect based on fundamental reaction[J]. China Safety Science Journal, 2024, 34(3): 101-108.
doi: 10.16265/j.cnki.issn1003-3033.2024.03.1169
|
| [18] |
ZHAO Tenglong, CHEN Xiaokun, LUO Zhenmin, et al. Effect of N 2 inerting on the inhibition of methane explosions by a multicomponent powder[J]. Fuel, 2023, 337(4): DOI: 10.1016/j.fuel.2022.127203.
|